
News - Page 1
Brain Awareness Week 2024

March 12, 2024
As every year, events for the general public are being held in around 100 countries to celebrate Brain Awareness Week. This year, events are taking place from March 11 to 17.
Members of the general public are invited to take part in a range of activities (meeting researchers, participating in workshops, attending conferences, etc.) to learn more about the brain and find out about advances in neuroscience research.
Read moreRare Disease Day 2024

February 29, 2024
Like every year on the last day of February, Santé Active Edition – Synergy Pharm is helping to raise awareness about rare diseases. On this Rare Disease Day, our medical writing team would like to express their support for patients, their families and friends, and all those involved in the fight against these diseases.
Read moreACCORD, a new reporting guideline

February 15, 2024
In the field of biomedical research, consensus between different professionals is often sought when developing clinical guidelines. The new ACCORD (ACcurate COnsensus Reporting Document) reporting guideline will enable the scientific community, and in particular medical writing professionals, to report accurately, comprehensively and consistently on the methods used, as well as the results obtained, within the framework of a consensus.
Read moreWorld Neglected Tropical Diseases Day 2024

January 29, 2024
Every year on 30 January, the World Health Organization (WHO) raises awareness of Neglected tropical diseases (NTD) as part of World NTD Day.
What are neglected tropical diseases?
Neglected tropical diseases (NTDs) are a group of infectious diseases that are caused by pathogens such as viruses, bacteria, parasites, fungi, and toxins and lead to an impaired health status or death if not treated. These diseases are mostly found in very poor communities living in tropical areas, and their etiology is often associated with specific environmental factors. While some NTDs can be eradicated, meaning that their incidence is reduced to zero permanently, those with animal or insect vectors can only be eliminated, meaning that human transmission is stopped locally, and then controlled to limit outbreaks.
Read moreClinical Trials Quebec: the new provincial resource for all things clinical research!

January 16, 2024
Santé Active Edition – Pharm is delighted to share the launch of Clinical Trials Quebec (CTQ)! Developed by the CATALIS Network, CTQ is a brand-new information and services hub for all things clinical research, supported by the Quebec government.
Read moreProtection from skin damage caused by high-energy visible light (HEV), also known as blue light

December 21, 2023
What is blue light?
Blue light is located at the edge of invisible ultraviolet (UV) light spectrum. Light with wavelengths in this part of the high-energy spectrum (400–450 nm) is also called high-energy visible (HEV) light.
What are the effects of HEV light on the skin?
In recent years, several studies have shown that skin damage caused by the sun is not only due to UV radiation. Visible light is more abundant and penetrates deeper into the skin layers than UV radiation. The damaging effects of visible light contribute to premature photoaging of the skin, lead to hyperpigmentation, and play a role in carcinogenesis.
Despite increasing exposure to artificial blue light sources (light-emitting diodes (LEDs), smartphone screens, computer screens, etc.), 95% to 99% of the harmful effects of blue light on the skin are due to the sun. It is therefore necessary to protect the skin from the whole spectrum of solar radiation.
Read moreTeam meeting in Paris

November 23, 2023
The Santé Active Edition – Synergy Pharm medical writing agency recently met up in Paris for a team seminar.
The meeting gave us the opportunity to review our activities over the past year, and discuss our various current and future medical and scientific writing projects.
Read moreFocus on the clients of Santé Active Edition – Synergy Pharm
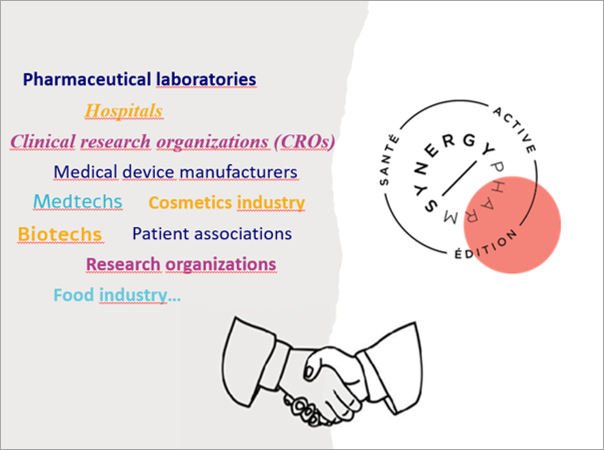
November 10, 2023
Santé Active Edition – Synergy Pharm meets the different needs of its clients thanks to:
- Santé Active Edition, which offers a variety of scientific and medical communication services for professionals and the general public (articles and reviews for international peer-reviewed journals, slideshows, posters and scientific congress proceedings, product brochures, patient brochures, medical or scientific sketchnotes, content for patient websites, books, etc.), and editorial assistance services (assistance with publication, basic to expert proofreading as needed, submission to peer-reviewed journals including the preparation of all required documents, point-by-point response letters to reviewers’ comments, translation of scientific texts, scientific coordination, etc.);
- Synergy Pharm, which is specialized in regulatory and clinical documentation (drafting of clinical study protocols, investigator brochures, clinical study reports, medical device evaluation reports, Common Technical Document (CTD) dossiers for drug registration, pharmacovigilance documents, answers to questions from regulatory authorities, etc.);
- The complementarity and adaptability of our qualified, experienced, rigorous, and proactive medical writing team.
Synergy Pharm has obtained Research Tax Credit (CIR) approval as a service provider carrying out research and development (R&D) work on behalf of its clients. Our clients who are eligible for the CIR can therefore benefit from this incentive.
Read moreA focus on Santé Active Edition: specialists in medical and scientific communication for professionals and the general public

October 27, 2023
The Santé Active Edition arm of the Santé Active Edition – Synergy Pharm health communication agency has been specializing in medical and scientific communication for professionals (researchers, physicians, veterinarians, pharmacists, etc.), patients, and the general public since 1998.
Read morePink October 2023

September 29, 2023
Pink October is the month-long annual campaign dedicated to raising awareness of breast cancer. As happens every year, Pink October 2023 will be marked by a large number of events organized to raise funds for medical and scientific research and increase awareness of the importance of early detection of breast cancer. In France, The Ruban Rose association is organizing an unprecedented event on Sunday the 1st of October on the Champs-Élysées for the launch of the 2023 Pink October campaign. In the UK, Breast Cancer UK is encouraging everyone to get active with a 30-day challenge to reduce the risk of developing breast cancer and raise money to support breast cancer prevention. In the US, the focus of the National Breast Cancer Foundation is on ensuring Pink October does “more than raise awareness” and promotes action on fundraising and screening.
Read more